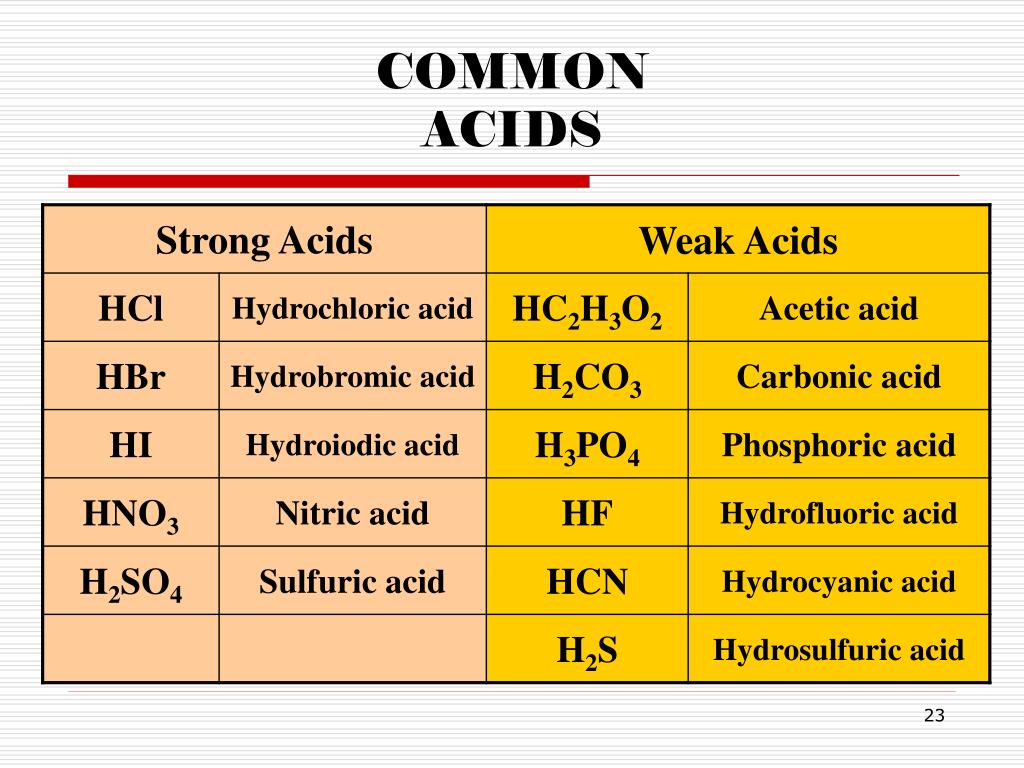
Chemical Properties of Acids:Īcids are chemicals that release hydrogen ions (H+) when dissolved in water. The boiling point is also high because the ions are strongly attracted to each other and are not able to move about as freely as they are in a liquid. This strong attraction between the ions holds the solid together, so the melting point is high. 
This is because the negatively charged ions in a base are attracted to each other, and this attraction increases as the temperature increases. The melting and boiling points of bases are higher than the melting and boiling points of acids. Bases are soluble in water, but insoluble in most organic solvents. They are odorless, but some can have a bitter taste. 
They are usually white or colorless, but can also be colored. The physical properties of bases are very different from the physical properties of acids.īases are often solids, although some are liquids. They are usually ionic compounds, but can also be covalent compounds. Physical properties of basesĪ base is a substance that can accept protons from acids to form salts. Some common bases include sodium hydroxide (NaOH), potassium hydroxide (KOH), and ammonium hydroxide (NH 4 OH). They are soluble in water and can form ionic compounds with acids. Bases are also electrolytes, which means that they can conduct electricity in solution.īases have a pH greater than 7.0 and are often referred to as alkalis. This means that a base can dissolve in water and release a hydroxide ion (OH-). A base is a substance that can accept a proton (H+) in an aqueous solution.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
